
Introduction
Pharmaceutical companies operate under some of the most demanding conditions of any industry. Between strict FDA oversight, massive clinical trial datasets, complex global supply chains, and zero tolerance for manufacturing errors, the pressure to maintain speed, accuracy, and compliance is relentless. Manual processes—spreadsheet reconciliations, paper-based batch record reviews, email-driven regulatory submissions—can't keep pace with the scale and complexity of modern pharmaceutical operations.
Robotic process automation (RPA) is emerging as a direct response to this pressure. The RPA in Healthcare market was valued at $1.5 billion in 2023 and is projected to reach $14 billion by 2032, growing at 13% annually. Pharmaceutical companies are deploying software bots across the entire value chain—from drug discovery and clinical trial management through manufacturing quality control and regulatory submissions.
Those that haven't started automating are already seeing the gap widen: longer submission timelines, more audit findings, and slower time-to-market compared to competitors who have.
This article covers where RPA creates measurable value in pharma—from compliance-specific demands like 21 CFR Part 11 and GMP to real-world outcomes—and what it takes to build a scalable automation program that sticks beyond a pilot.
TLDR
- RPA automates rule-based, high-volume tasks across clinical trials, manufacturing, supply chain, and regulatory submissions
- Key use cases include adverse event reporting, batch record generation, and regulatory document preparation — areas where speed and accuracy directly affect compliance outcomes
- Cuts human error and reporting timelines while generating audit-ready data trails that satisfy FDA and GMP requirements
- Always start with process assessment, then pilot, then scale — skipping steps is how implementations fail
- ROI extends beyond cost savings to faster compliance cycles, reduced audit risk, improved data quality
What Is RPA in Pharmaceutical Manufacturing?
RPA refers to software bots that mimic human actions on computer systems—logging into applications, extracting data from forms, filling regulatory templates, transferring records between databases, and triggering workflows. These bots operate without requiring custom API integrations or changes to existing infrastructure, which matters in pharma where legacy systems are common and change control processes are strict.
The pharmaceutical industry is an exceptionally strong fit for RPA because it generates enormous volumes of structured, rules-based data. Clinical trial records, batch manufacturing logs, regulatory filings, procurement transactions—all follow predictable patterns and formats. This is exactly where RPA performs best: high-volume, repetitive, rule-governed tasks that don't require complex judgment but demand perfect consistency.
One clarification before going further: this article covers software-based process automation, not physical robotics like robotic arms on manufacturing lines. RPA acts as a connective layer between legacy systems that don't otherwise communicate, automatically moving data without costly custom integration projects. That includes systems such as:
- ERP platforms — for procurement, inventory, and financial workflows
- LIMS (Laboratory Information Management Systems) — for lab data capture and reporting
- QMS (Quality Management Systems) — for deviation tracking, CAPA, and audit trails
Where RPA Delivers Value Across the Pharma Value Chain
RPA's impact isn't isolated to one department—it spans the entire pharmaceutical value chain. This section maps the highest-value automation opportunities by operational stage.
Drug Discovery and Clinical Trial Management
RPA accelerates drug discovery by automating high-throughput data collection, patient matching for trial eligibility, and aggregation of experimental results from multiple sources into centralized databases. Instead of research teams manually compiling results from different lab systems, bots pull data automatically and flag anomalies in real time.
Clinical trial automation tasks include:
- Automated data entry from electronic health records into trial databases
- Query resolution tracking across sites and investigators
- Trial supply inventory updates and shipment notifications
- Monitoring report generation for safety review boards
Pharmacovigilance (PV) case processing is a critical bottleneck. Case processing activities consume approximately 67% of a pharmaceutical company's internal PV resource use and most of the overall PV budget when outsourcing costs are included.
Research from Pfizer showed that AI and RPA together could process approximately 33% of adverse event cases to 80%+ completeness — cutting manual workload on safety teams without sacrificing data quality.

Manufacturing, Quality Control, and Batch Records
RPA automates the generation and verification of batch manufacturing records (BMRs), cross-referencing actual production data against specifications and flagging deviations automatically. This reduces the hours teams spend on manual review and eliminates transcription errors that can trigger costly batch failures or regulatory findings.
Quality control automation:
- Pull inspection data from lab instruments and manufacturing execution systems
- Run pass/fail comparisons against GMP thresholds
- Generate quality reports in standardized formats
- Route exceptions to the appropriate reviewer without human involvement in routine checks
This eliminates human variability, enforces process consistency, and creates timestamped audit trails that regulators require.
These same traceability requirements extend downstream — making supply chain operations another high-priority automation target.
Supply Chain, Inventory, and Procurement
RPA handles inventory monitoring across multiple distribution points, auto-generates replenishment orders when stock thresholds are met, and reconciles purchase orders with incoming shipments. This reduces stockout risk and overstocking costs while ensuring serialization and traceability compliance.
Rebate and chargeback processing is one of the highest-risk revenue leakage points in pharma. KPMG estimates that fraudulent copay/coupon claims cost manufacturers tens of millions of dollars per year — in one test case, four pharmacies collected approximately $1 million in fraudulent reimbursements in 2018 alone.
RPA automates data collection and validation across multiple interfaces, flagging duplicate rebates, chargeback abuse, and copay misuse before payments are issued.
Back-office automation includes:
- Automates supplier onboarding — candidate research, invite workflows, and ERP vendor master creation
- Runs three-way matching of PO, receipt, and invoice for faster, error-free invoice processing
- Posts GL entries automatically and flags discrepancies for finance review
For most pharma operations teams, back-office automation is where RPA deployments start — the ROI is measurable within the first quarter and the scope is contained enough to build internal confidence before scaling.
RPA and Regulatory Compliance: Meeting FDA, GMP, and DSCSA Standards
Compliance is not optional in pharma, and manual compliance processes are a liability. Many regulatory tasks—document gathering, data verification, submission formatting—are rules-based and therefore well-suited to RPA.
Adverse Event and Pharmacovigilance Reporting
Bots aggregate AE data from multiple sources—clinical databases, call centers, social media monitoring tools—validate fields against submission requirements, and auto-populate regulatory forms for FDA MedWatch or EMA submissions. This reduces reporting cycle time and eliminates the risk of late or inaccurate filings that trigger warning letters.
FDA 21 CFR Part 11 Compliance
Section 11.10(e) mandates secure, computer-generated, time-stamped audit trails that independently record when operators create, modify, or delete electronic records. RPA creates tamper-evident audit trails of every automated action—who ran the bot, what data it accessed, when, and what it changed. This improves audit readiness over manual processes, where documentation is often incomplete.
GMP Compliance
RPA enforces process consistency by executing steps in the same sequence every time. Unlike human operators who may skip steps or perform tasks out of order, bots eliminate the variability that leads to GMP deviations. This covers three high-risk documentation areas:
- Cleanroom entry and exit logs
- Batch record completeness and accuracy
- Equipment cleaning and verification sign-offs
DSCSA and Drug Supply Chain Traceability
The Drug Supply Chain Security Act requires pharmaceutical companies to build interoperable systems that track prescription drugs across distribution networks. RPA supports this by:
- Monitoring and tracing serialized products across the supply chain
- Verifying supplier authenticity against FDA databases
- Triggering investigation workflows when potentially illegitimate drugs are detected
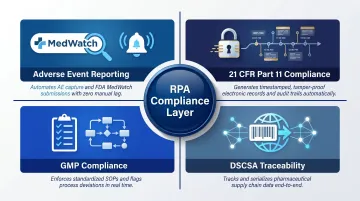
Key Benefits Pharma Companies Gain from RPA
Accuracy and Error Reduction
RPA eliminates data entry errors, transcription mistakes, and process inconsistencies. 90% of organizations reported improved quality and accuracy after RPA implementation, according to Deloitte's global RPA survey. In a hospital case study, manual operations time was reduced from 34.3 minutes to 3.4 minutes per person—a 90% reduction in waste, with process cycle efficiency improving from 69% to 96%.
In pharma, errors have serious consequences: regulatory penalties, patient safety risks, batch failures, and delayed product launches. Automating high-risk data processes keeps those risks out of human hands entirely.
Speed and Throughput
RPA enables 24/7 operation without fatigue. Unlike human operators, bots run continuously — no shift changes, no fatigue-related errors. That consistency compresses reporting cycles and accelerates trial data processing at scale.
One EY report found that RPA reduced form processing time from 2 hours to 3 minutes. For monthly regulatory submissions that involve hundreds of forms, this scales to massive time savings.
Cost Reduction and ROI
Deloitte's research indicates that organizations initially target 59% cost reduction from RPA but typically realize 20-40% in early stages, with most organizations achieving payback in under 12 months. RPA typically provides a 20% increase in FTE capacity without adding headcount.
Cost benefits extend beyond labor savings:
- Reduced rework costs from error correction
- Faster audit preparation (hours instead of weeks)
- Lower compliance penalty risk
- Reduced supply chain inefficiency and revenue leakage
For pharma companies running dozens of repetitive data workflows, those savings add up fast — often before the first year of deployment is complete.
How to Build a Scalable RPA Program in Pharma
Step 1 — Process Assessment and Prioritization
Start by identifying high-volume, rules-based processes with clear inputs and outputs. Avoid starting with your most complex workflows. Conduct a process audit using this framework:
Ideal RPA candidates are processes that are:
- Repetitive (performed dozens or hundreds of times per week)
- Prone to human error (data entry, copy/paste operations)
- Time-consuming (consuming significant FTE hours)
- Involve structured data (forms, databases, or spreadsheets)
- Rule-based (follow a defined logic without requiring complex judgment)

Don't start with the hardest problem. Start where you'll see fast, measurable results.
Step 2 — Proof of Concept (Pilot)
Select a small, well-defined process for your first RPA deployment. The goal is to prove value quickly, gather performance data, and build internal confidence before scaling.
Critical pilot requirements:
- Set measurable goals before launch (cycle time reduction, error rate, hours saved)
- Choose a process that's painful enough to matter but simple enough to automate quickly
- Involve the process owners — frontline staff who know where the pain points are
- Document baseline metrics so you can quantify improvement
A well-scoped pilot can go live in 4-8 weeks.
Step 3 — Selecting the Right RPA Platform for Pharma
Choose your RPA platform based on pharma-specific criteria:
- Auditability and logging: Does it support 21 CFR Part 11 compliance with timestamped, tamper-evident audit trails?
- Integration capability: Can it connect to LIMS, ERP, QMS, and other pharma-specific systems?
- Scalability: Will it support enterprise-wide deployment across multiple geographies and business units?
- Vendor support: Does the vendor understand regulated industry requirements?
Common RPA platforms in pharma include UiPath, Automation Anywhere, and Blue Prism, all recognized as leaders in the 2025 Gartner Magic Quadrant for RPA. Selection depends on your organization's existing technology stack, compliance requirements, and scale ambitions.
Step 4 — Change Management and Workforce Transition
RPA implementations fail when organizations treat them as purely IT projects. Successful automation requires clear communication about bot roles versus human roles, reskilling programs, and involving frontline staff in process mapping (not just IT).
Address workforce resistance directly:
- Explain that automation typically elevates roles (from manual data entry to bot monitoring and exception handling) rather than eliminating them
- Involve process owners in design so they feel ownership, not threatened
- Provide training on how to work alongside bots
- Celebrate quick wins publicly
Automation should free people for higher-value work: clinical judgment, exception handling, and strategic analysis. When staff see that shift happening, scaling becomes far easier to justify.
Step 5 — Scale and Continuous Improvement
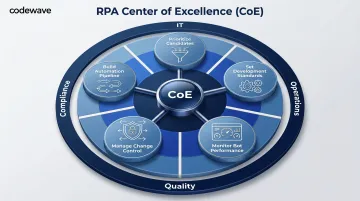
Scaling RPA from one process to an enterprise-wide program requires a Center of Excellence (CoE). This cross-functional team (IT, operations, quality, compliance) governs bot deployment, monitors performance, and identifies new automation candidates.
CoE responsibilities include:
- Prioritizing new automation candidates
- Setting standards for bot development and testing
- Monitoring bot performance and uptime
- Managing change control and validation for regulated processes
- Building a pipeline of automation opportunities
Only 3% of organizations have scaled their digital workforce to more than 50 robots, primarily due to process fragmentation, IT readiness issues, and cultural pushback. A strong CoE overcomes these barriers.
An experienced implementation partner can shorten that path significantly. Codewave's ImpactIndex™ model ties engagement fees to measurable outcomes rather than hours logged, which reduces the risk of stalled pilots. With implementations across 400+ businesses, including regulated industries, Codewave supports pharma teams from initial process assessment through full CoE buildout.
Frequently Asked Questions
What is RPA in the pharmaceutical industry?
RPA refers to software bots that automate repetitive, rules-based digital tasks across pharma operations—such as data entry, report generation, regulatory submissions, and supply chain management—without replacing existing systems or requiring physical robots.
What are the top RPA software tools used in the pharmaceutical industry?
The most widely adopted RPA platforms in pharma include UiPath, Automation Anywhere, Blue Prism, and SAP Intelligent RPA. Selection should be based on compliance logging capabilities, integration with LIMS/ERP/QMS, and scalability requirements for your environment.
Which pharmaceutical processes are best suited for RPA automation?
The highest-ROI starting points are adverse event reporting, batch record generation, inventory replenishment, regulatory document preparation, clinical trial data entry, and invoice processing.
How does RPA support FDA regulatory compliance in pharma?
RPA creates consistent, timestamped audit trails for every automated action (supporting 21 CFR Part 11), reduces errors in regulatory submissions, enforces GMP process consistency, and accelerates adverse event reporting timelines.
What ROI can pharma companies expect from RPA implementation?
ROI varies by process but commonly includes 20-40% cost reduction in targeted workflows, 90%+ accuracy improvement, and faster processing times—hours instead of days. Define measurable KPIs before implementation to track actual returns.
How long does it take to implement RPA in a pharmaceutical company?
A well-scoped pilot can go live in 4-8 weeks, while enterprise-scale deployment across multiple processes typically takes 6-18 months depending on system complexity, validation requirements, and change management needs.


